Note
This page was generated from sccoda_extended.ipynb. Some tutorial content may look better in light mode.
scCODA - Modeling options and result analysis¶
This tutorial notebook serves as an extension to the general tutorial and presents ways to alternate the model and perform more in-depth result analysis and diagnostics. We will focus on:
Modifications of the model formula and reference cell type to perform different modeling tasks
Plotting of compositional data and scCODA results
Inference methods available in scCODA
Advanced interpretation and analysis of results
Alternative differential abundance testing using all references
We will again analyze the small intestinal epithelium data of mice from Haber et al., 2017. First, we read in the data and perform the same preprocessing steps as in the general tutorial:
[1]:
import warnings
warnings.filterwarnings("ignore")
import arviz as az
import matplotlib.pyplot as plt
import pandas as pd
import pertpy as pt
[2]:
# Load data
haber_cells = pt.dt.haber_2017_regions()
# Convert data to mudata object
sccoda_model = pt.tl.Sccoda()
sccoda_data = sccoda_model.load(
haber_cells,
type="cell_level",
generate_sample_level=True,
cell_type_identifier="cell_label",
sample_identifier="batch",
covariate_obs=["condition"],
)
# Select control and salmonella data as one modality
sccoda_data.mod["coda_salm"] = sccoda_data["coda"][
sccoda_data["coda"].obs["condition"].isin(["Control", "Salmonella"])
].copy()
print(sccoda_data)
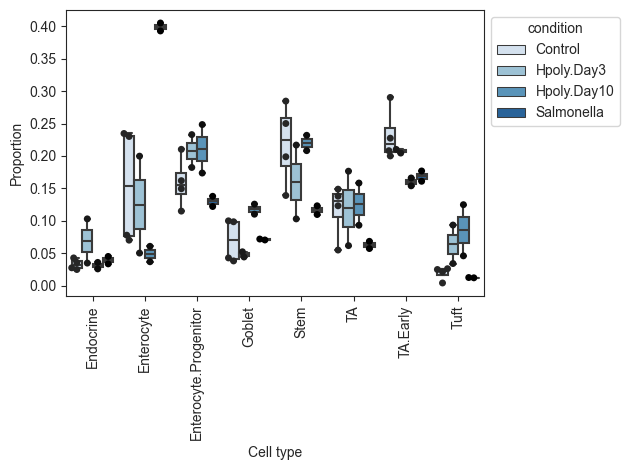
# plot boxplots
pt.pl.coda.boxplots(sccoda_data, modality_key="coda", feature_name="condition", add_dots=True)
plt.show()
MuData object with n_obs × n_vars = 9852 × 15223
3 modalities
rna: 9842 x 15215
obs: 'batch', 'barcode', 'condition', 'cell_label'
coda: 10 x 8
obs: 'condition'
var: 'n_cells'
coda_salm: 6 x 8
obs: 'condition'
var: 'n_cells'
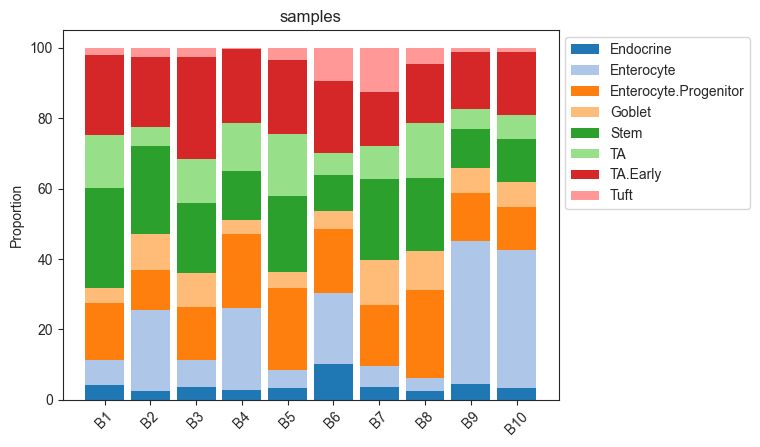
Alternatively, we can also plot the data as stacked barplots:
[3]:
# Stacked barplot for each sample
pt.pl.coda.stacked_barplot(sccoda_data, modality_key="coda", feature_name="samples")
plt.show()
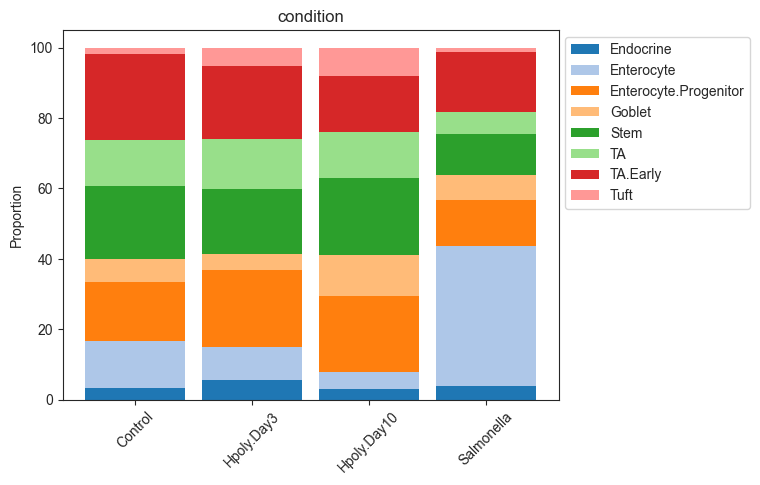
# Stacked barplot for the levels of "Condition"
pt.pl.coda.stacked_barplot(sccoda_data, modality_key="coda", feature_name="condition")
plt.show()
Tweaking the model formula and reference cell type¶
First, we take a closer look at how changing the formula parameter of the scCODA model influences the results. Internally, the formula string is converted into a linear model-like design matrix via patsy, which has a similar syntax to the lm function in the R language.
Multi-level categories¶
Patsy allows us to automatically handle categorical covariates, even with multiple levels. For example, we can model the effect of all three diseases at once, using the coda data modality, which contains all 10 samples:
[4]:
# model all three diseases at once
sccoda_data = sccoda_model.prepare(
sccoda_data,
modality_key="coda",
formula="condition",
reference_cell_type="Endocrine",
)
sccoda_model.run_nuts(sccoda_data, modality_key="coda")
sccoda_model.summary(sccoda_data, modality_key="coda")
sample: 100%|██████████| 11000/11000 [01:05<00:00, 166.83it/s, 127 steps of size 2.09e-02. acc. prob=0.75]
Compositional Analysis summary ┌─────────────────────────────────────────────┬───────────────────────────────────────────────────────────────────┐ │ Name │ Value │ ├─────────────────────────────────────────────┼───────────────────────────────────────────────────────────────────┤ │ Data │ Data: 10 samples, 8 cell types │ │ Reference cell type │ Endocrine │ │ Formula │ condition │ └─────────────────────────────────────────────┴───────────────────────────────────────────────────────────────────┘
┌─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┐ │ Intercepts │ ├─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┤ │ Final Parameter Expected Sample │ │ Cell Type │ │ Endocrine 0.970 46.752 │ │ Enterocyte 1.893 117.666 │ │ Enterocyte.Progenitor 2.328 181.790 │ │ Goblet 1.458 76.161 │ │ Stem 2.399 195.166 │ │ TA 1.857 113.505 │ │ TA.Early 2.519 220.049 │ │ Tuft 0.625 33.111 │ └─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┘
┌─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┐ │ Effects │ ├─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┤ │ Final Parameter Expected Sample log2-fold change │ │ Covariate Cell Type │ │ conditionT.Hpoly.Day3 Endocrine 0.000 46.752 0.000 │ │ Enterocyte 0.000 117.666 0.000 │ │ Enterocyte.Progenitor 0.000 181.790 0.000 │ │ Goblet 0.000 76.161 0.000 │ │ Stem 0.000 195.166 0.000 │ │ TA 0.000 113.505 0.000 │ │ TA.Early 0.000 220.049 0.000 │ │ Tuft 0.000 33.111 0.000 │ │ conditionT.Hpoly.Day10 Endocrine 0.000 46.752 0.000 │ │ Enterocyte 0.000 117.666 0.000 │ │ Enterocyte.Progenitor 0.000 181.790 0.000 │ │ Goblet 0.000 76.161 0.000 │ │ Stem 0.000 195.166 0.000 │ │ TA 0.000 113.505 0.000 │ │ TA.Early 0.000 220.049 0.000 │ │ Tuft 0.000 33.111 0.000 │ │ conditionT.Salmonella Endocrine 0.000 32.433 -0.528 │ │ Enterocyte 1.546 383.065 1.703 │ │ Enterocyte.Progenitor 0.000 126.112 -0.528 │ │ Goblet 0.000 52.835 -0.528 │ │ Stem 0.000 135.391 -0.528 │ │ TA 0.000 78.741 -0.528 │ │ TA.Early 0.000 152.653 -0.528 │ │ Tuft 0.000 22.970 -0.528 │ └─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┘
Different reference levels¶
Per default, categorical variables are encoded via full-rank treatment coding. Hereby, the value of the first sample in the dataset is used as the default (control) category. We can select the default level by changing the model formula to "C(<CovariateName>, Treatment('<ReferenceLevelName>'))":
For example, we can switch the salmonella model to test diseased versus healthy samples, which switches the sign of the only credible effect (Enterocytes).
[22]:
# Set salmonella infection as "default" category
sccoda_data = sccoda_model.prepare(
sccoda_data,
modality_key="coda_salm",
formula="C(condition, Treatment('Salmonella'))",
reference_cell_type="Goblet",
)
sccoda_model.run_nuts(sccoda_data, modality_key="coda_salm")
sccoda_model.summary(sccoda_data, modality_key="coda_salm")
sample: 100%|██████████| 11000/11000 [00:47<00:00, 230.14it/s, 255 steps of size 1.80e-02. acc. prob=0.86]
Compositional Analysis summary ┌────────────────────────────────────────┬────────────────────────────────────────────────────────────────────────┐ │ Name │ Value │ ├────────────────────────────────────────┼────────────────────────────────────────────────────────────────────────┤ │ Data │ Data: 6 samples, 8 cell types │ │ Reference cell type │ Goblet │ │ Formula │ C(condition, Treatment('Salmonella')) │ └────────────────────────────────────────┴────────────────────────────────────────────────────────────────────────┘
┌─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┐ │ Intercepts │ ├─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┤ │ Final Parameter Expected Sample │ │ Cell Type │ │ Endocrine 1.229 28.334 │ │ Enterocyte 3.670 325.399 │ │ Enterocyte.Progenitor 2.541 105.220 │ │ Goblet 1.746 47.515 │ │ Stem 2.594 110.947 │ │ TA 2.009 61.809 │ │ TA.Early 2.849 143.173 │ │ Tuft 0.419 12.604 │ └─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┘
┌─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┐ │ Effects │ ├─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┤ │ Final Parameter Expected Sample │ │ log2-fold change │ │ Covariate Cell Type │ │ C(condition, Treatment('Salmonella'))T.Control Endocrine 0.000 39.776 │ │ 0.489 │ │ Enterocyte -1.340 119.591 │ │ -1.444 │ │ Enterocyte.Progenitor 0.000 147.714 │ │ 0.489 │ │ Goblet 0.000 66.705 │ │ 0.489 │ │ Stem 0.000 155.754 │ │ 0.489 │ │ TA 0.000 86.771 │ │ 0.489 │ │ TA.Early 0.000 200.994 │ │ 0.489 │ │ Tuft 0.000 17.695 │ │ 0.489 │ └─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┘
Switching the reference cell type¶
Compositional analysis generally does not allow statements on absolute abundance changes, but only in relation to a reference category, which is assumed to be unchanged in absolute abundance. The reference cell type fixes this category in scCODA. Thus, an interpretation of scCODA’s effects should always be formulated like:
“Using cell type xy as a reference, cell types (a, b, c) were found to credibly change in abundance”
Switching the reference cell type might thus produce different results. For example, if we choose a different cell type as the reference (such as Enterocytes in the salmonella infection data), scCODA can find other credible effects on the other cell types.
Note: In this example we have to set the FDR very high, since all cell types except Enterocytes do not show big changes. FDR values of more than 0.2 are not recommended in practice
[23]:
sccoda_data = sccoda_model.prepare(
sccoda_data,
modality_key="coda_salm",
formula="condition",
reference_cell_type="Enterocyte",
)
sccoda_model.run_nuts(sccoda_data, modality_key="coda_salm")
sccoda_model.summary(sccoda_data, modality_key="coda_salm", est_fdr=0.4)
sample: 100%|██████████| 11000/11000 [00:32<00:00, 340.52it/s, 63 steps of size 6.98e-02. acc. prob=0.70]
Compositional Analysis summary ┌──────────────────────────────────────────────┬──────────────────────────────────────────────────────────────────┐ │ Name │ Value │ ├──────────────────────────────────────────────┼──────────────────────────────────────────────────────────────────┤ │ Data │ Data: 6 samples, 8 cell types │ │ Reference cell type │ Enterocyte │ │ Formula │ condition │ └──────────────────────────────────────────────┴──────────────────────────────────────────────────────────────────┘
┌─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┐ │ Intercepts │ ├─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┤ │ Final Parameter Expected Sample │ │ Cell Type │ │ Endocrine 0.550 34.807 │ │ Enterocyte 2.075 159.941 │ │ Enterocyte.Progenitor 1.861 129.128 │ │ Goblet 1.089 59.669 │ │ Stem 2.086 161.710 │ │ TA 1.472 87.515 │ │ TA.Early 2.211 183.242 │ │ Tuft -0.056 18.988 │ └─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┘
┌─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┐ │ Effects │ ├─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┤ │ Final Parameter Expected Sample log2-fold change │ │ Covariate Cell Type │ │ condition[T.Salmonella] Endocrine 0.000 41.619 0.258 │ │ Enterocyte 0.000 191.246 0.258 │ │ Enterocyte.Progenitor 0.000 154.402 0.258 │ │ Goblet 0.000 71.347 0.258 │ │ Stem -0.468 121.082 -0.417 │ │ TA -0.380 71.579 -0.290 │ │ TA.Early -0.308 161.019 -0.187 │ │ Tuft 0.000 22.704 0.258 │ └─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┘
Finding a reference cell type¶
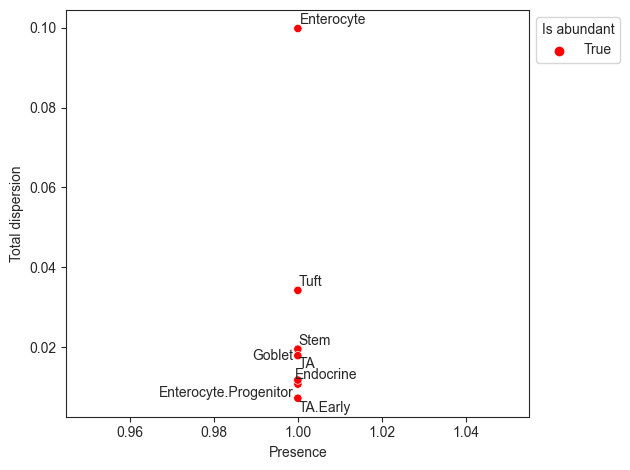
The scCODA model requires a cell type to be set as the reference category. However, choosing this cell type is often difficult. A good first choice is a referenece cell type that closely preserves the changes in relative abundance during the compositional analysis.
For this, it is important that the reference cell type is not rare, to avoid large relative changes being caused by small absolute changes. Also, the relative abundance of the reference should vary as little as possible across all samples.
The visualization rel_abundance_dispersion_plot shows the presence (share of non-zero samples) over all samples for each cell type versus its dispersion in relative abundance. Cell types that have a higher presence than a certain threshold (default 0.9) are suitable candidates for the reference and thus colored.
[24]:
pt.pl.coda.rel_abundance_dispersion_plot(sccoda_data, modality_key="coda", abundant_threshold=0.9)
[24]:
<AxesSubplot: xlabel='Presence', ylabel='Total dispersion'>
Inference algorithms in scCODA¶
scCODA performs parameter inference via Markov-chain Monte Carlo (MCMC) methods. There are three different MCMC sampling methods available for scCODA:
HMC sampling with Dual-averaging step size adaptation (Nesterov, 2009):
run_hmc()No-U-Turn sampling (Hoffman and Gelman, 2014):
run_nuts()
Generally, it is recommended to use NUTS sampling. Other methods, such as variational inference, are in consideration.
For all MCMC sampling methods, properties such as the MCMC chain length and the number of burn-in samples are directly adjustable.
Result analysis and diagnostics¶
The “getting started” tutorial explains how to do interpret the basic output of scCODA. To follow this up, we now take a look at how MCMC diagnostics and more advanced result analysis in scCODA can be performed.
For this section, we again use the model of salmonella infection versus control group, with a reference cell type of Goblet cells.
[25]:
sccoda_data = sccoda_model.prepare(
sccoda_data,
modality_key="coda_salm",
formula="condition",
reference_cell_type="Goblet",
)
sccoda_model.run_nuts(sccoda_data, modality_key="coda_salm")
sample: 100%|██████████| 11000/11000 [00:39<00:00, 275.96it/s, 255 steps of size 2.61e-02. acc. prob=0.82]
Extended model summary¶
sccoda_model.summary() can give us, apart from the properties already explained in the basic tutorial, more information about the posterior inferred by the model by setting extended=True. The extended summary also includes some information on the MCMC sampling procedure (chain length, burn-in, acceptance rate, duration).
For both effects and intercepts, we also get the standard deviation (SD) and high density interval endpoints of the posterior density of the generated Markov chain.
The effects summary also includes the spike-and-slab inclusion probability for each effect, i.e. the share of MCMC samples, for which this effect was not set to 0 by the spike-and-slab prior. A threshold on this value serves as the deciding factor whether an effect is considered statistically credible
We can also use the summary tables from summary() as pandas DataFrames to tweak them further. They are also accessible via sccoda_model.get_intercept_df() and sccoda_model.get_effect_df(), respectively. Furthermore, the tables are generated through the summary() function in arviz and support all its functionality. This means that we can, for example, easily change the credible interval of parameters:
[26]:
sccoda_model.summary(sccoda_data, modality_key="coda_salm", hdi_prob=0.8, extended=True)
Compositional Analysis summary ┌───────────────────────────────────────┬─────────────────────────────────────────────────────────────────────────┐ │ Name │ Value │ ├───────────────────────────────────────┼─────────────────────────────────────────────────────────────────────────┤ │ Data │ Data: 6 samples, 8 cell types │ │ Reference cell type │ Goblet │ │ Formula │ condition │ │ Reference index │ 3 │ │ Spike-and-slab threshold │ 1.000 │ │ Spike-and-slab threshold │ 1.000 │ │ MCMC Sampling │ Sampled 10000 chain states (1000 burnin samples) │ │ Acceptance rate │ 81.8% │ └───────────────────────────────────────┴─────────────────────────────────────────────────────────────────────────┘
┌─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┐ │ Intercepts │ ├─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┤ │ Final Parameter HDI 10% HDI 90% SD Expected Sample │ │ Cell Type │ │ Endocrine 1.118 0.699 1.590 0.357 34.667 │ │ Enterocyte 2.329 1.961 2.741 0.310 116.372 │ │ Enterocyte.Progenitor 2.519 2.129 2.915 0.311 140.723 │ │ Goblet 1.747 1.355 2.158 0.321 65.027 │ │ Stem 2.703 2.335 3.101 0.304 169.152 │ │ TA 2.109 1.705 2.532 0.326 93.391 │ │ TA.Early 2.861 2.482 3.244 0.301 198.105 │ │ Tuft 0.438 -0.036 0.946 0.394 17.563 │ └─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┘
┌─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┐ │ Effects │ ├─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┤ │ Final Parameter Expected Sample log2-fold change │ │ Covariate Cell Type │ │ condition[T.Salmonella] Endocrine 0.000 24.752 -0.486 │ │ Enterocyte 1.354 321.913 1.468 │ │ Enterocyte.Progenitor 0.000 100.474 -0.486 │ │ Goblet 0.000 46.428 -0.486 │ │ Stem 0.000 120.771 -0.486 │ │ TA 0.000 66.680 -0.486 │ │ TA.Early 0.000 141.443 -0.486 │ │ Tuft 0.000 12.540 -0.486 │ └─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┘
┌─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┐ │ Effects Extended │ ├─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┤ │ HDI 10% HDI 90% SD Inclusion probability │ │ Covariate Cell Type │ │ condition[T.Salmonella] Endocrine -0.136 0.848 0.319 0.452 │ │ Enterocyte 1.010 1.676 0.263 1.000 │ │ Enterocyte.Progenitor -0.185 0.461 0.159 0.321 │ │ Goblet 0.000 0.000 0.000 0.000 │ │ Stem -0.589 0.040 0.212 0.388 │ │ TA -0.613 0.134 0.232 0.385 │ │ TA.Early -0.289 0.274 0.127 0.283 │ │ Tuft -0.506 0.658 0.308 0.398 │ └─────────────────────────────────────────────────────────────────────────────────────────────────────────────────┘
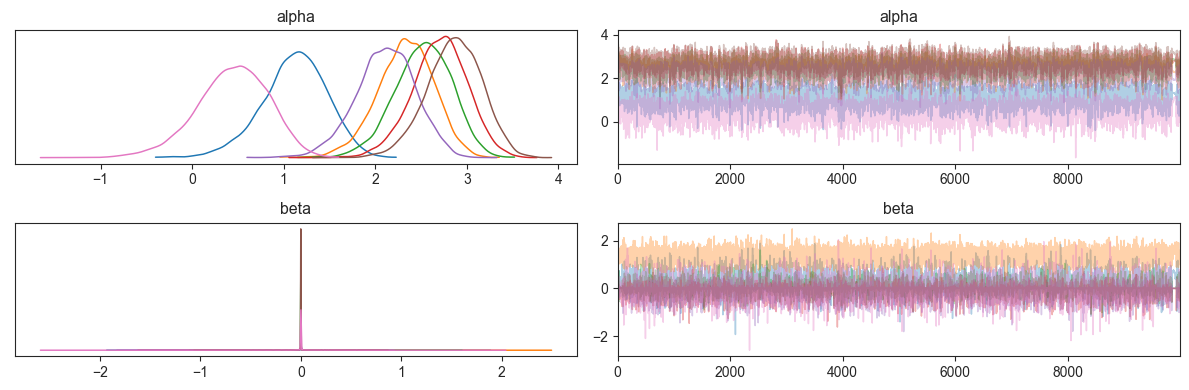
Diagnostics and plotting¶
Similarly to the summary dataframes being compatible with arviz, e can convert the result to an extension of arviz’s Inference Data class via make_arviz. This means that we can use all its MCMC diagnostic and plotting functionality. As an example, looking at the MCMC trace plots and kernel density estimates, we see that they are indicative of a well sampled MCMC chain:
Note: Due to the spike-and-slab priors, the beta parameters have many values at 0, which looks like a convergence issue, but is actually not.
Caution: Trying to plot a kernel density estimate for an effect on the reference cell type results in an error, since it is constant at 0 for the entire chain. To avoid this, add coords={"cell_type": salm_results.posterior.coords["cell_type_nb"]} as an argument to ``az.plot_trace``, which causes the plots for the reference cell type to be skipped.
[29]:
salm_arviz = sccoda_model.make_arviz(sccoda_data, modality_key="coda_salm")
az.plot_trace(
salm_arviz,
divergences=False,
var_names=["alpha", "beta"],
coords={"cell_type": salm_arviz.posterior.coords["cell_type_nb"]},
)
plt.tight_layout()
plt.show()
Using all cell types as reference alternatively to reference selection¶
scCODA uses a reference cell type that is considered to be unchanged over the experiment to guarantee the unique identifiability of results. If no such cell type is known beforehand, setting reference_cell_type="automatic" will find a suited reference. Alternatively, it is possible to find credible effects on cell types that are mostly independent of the reference. By sequentially running scCODA and selecting each cell type as the reference once, we can then use a majority vote to find the
cell types that were credibly changing more than half of the time.
Below, an example code for this procedure on the Salmonella infection data shows that only Enterocytes were found to be credible more than half of the time. Indeed, they are credibly changing for every reference cell type except themselves. All other cell types were not found to change with any reference.
[34]:
# Run scCODA with each cell type as the reference
cell_types = sccoda_data["coda_salm"].var.index
results_cycle = pd.DataFrame(index=cell_types, columns=["times_credible"]).fillna(0)
for ct in cell_types:
print(f"Reference: {ct}")
# Run inference
sccoda_data = sccoda_model.prepare(
sccoda_data,
modality_key="coda_salm",
formula="condition",
reference_cell_type=ct,
)
sccoda_model.run_nuts(sccoda_data, modality_key="coda_salm")
# Select credible effects
cred_eff = sccoda_model.credible_effects(sccoda_data, modality_key="coda_salm")
cred_eff.index = cred_eff.index.droplevel(level=0)
# add up credible effects
results_cycle["times_credible"] += cred_eff.astype("int")
Reference: Endocrine
sample: 100%|██████████| 11000/11000 [00:41<00:00, 264.03it/s, 255 steps of size 2.40e-02. acc. prob=0.83]
Reference: Enterocyte
sample: 100%|██████████| 11000/11000 [00:29<00:00, 375.11it/s, 31 steps of size 9.02e-02. acc. prob=0.63]
Reference: Enterocyte.Progenitor
sample: 100%|██████████| 11000/11000 [00:41<00:00, 264.37it/s, 127 steps of size 2.26e-02. acc. prob=0.87]
Reference: Goblet
sample: 100%|██████████| 11000/11000 [00:37<00:00, 294.89it/s, 127 steps of size 2.88e-02. acc. prob=0.75]
Reference: Stem
sample: 100%|██████████| 11000/11000 [00:43<00:00, 254.13it/s, 127 steps of size 2.02e-02. acc. prob=0.87]
Reference: TA
sample: 100%|██████████| 11000/11000 [00:36<00:00, 301.89it/s, 127 steps of size 3.26e-02. acc. prob=0.71]
Reference: TA.Early
sample: 100%|██████████| 11000/11000 [00:38<00:00, 282.39it/s, 127 steps of size 2.59e-02. acc. prob=0.81]
Reference: Tuft
sample: 100%|██████████| 11000/11000 [00:46<00:00, 238.00it/s, 255 steps of size 1.76e-02. acc. prob=0.90]
[35]:
# Calculate percentages
results_cycle["pct_credible"] = results_cycle["times_credible"] / len(cell_types)
results_cycle["is_credible"] = results_cycle["pct_credible"] > 0.5
print(results_cycle)
times_credible pct_credible is_credible
cell_label
Endocrine 0 0.000 False
Enterocyte 7 0.875 True
Enterocyte.Progenitor 0 0.000 False
Goblet 0 0.000 False
Stem 0 0.000 False
TA 0 0.000 False
TA.Early 0 0.000 False
Tuft 0 0.000 False